Technical Notes
This section presents technical notes explaining thermodynamic phenomena and provides recommendations on which methods and models to use to solve problems within fluid phase behavior.
Table of contents
- Quality Checking an Equation of State Model
- Allocation of Feed Streams to Oil & Gas Process Plant
- What Determines a Reservoir Fluid Composition?
- When to Use Non-Zero Binary Interaction Parameters in Equation of State Modeling
- 150 Years History of Cubic Equations of State
- What Causes Asphaltenes to Precipitate and How is it Modeled?
- Stages of Carbon Capture Storage
- EoS Modelling with the Critical Point as Anchor Point
Quality Checking an Equation of State Model
Most oil phases remain single-phase during production, but liquid-liquid splits can occur. This may for example be the case if the oil contains asphaltenes or has a high concentration of CO2. Such phase splits can be difficult to simulate with an equation of state (EoS) model, while on the other hand, it can also be difficult to ensure that an EoS model that matches experimental PVT data at the reservoir temperature does not simulate unphysical liquid-liquid splits at lower temperatures. Tc, Pc and acentric factor (ω) are well-defined quantities for lighter components but not for the C7+ components. It is the parameters a and b that enter in a cubic equation of state, not Tc, Pc and ω. This makes it difficult to establish rules for how Tc, Pc and ω of the C7+ components should develop with molecular weight. By shifting the focus from critical properties to the a and b parameters, it is possible to establish rules for how to develop an EoS model that simulates a liquid-liquid split when existing and otherwise not.
The van der Waals equation of state is shown in Figure 1 and relates pressure (P), molar volume (V), and temperature (T) of a pure component through two constants, a and b, both of which are unique functions of the critical temperature (Tc) and critical pressure (Pc). van der Waals developed the equation after realizing that the properties of a pure hydrocarbon throughout the PT range are essentially determined by the temperature and pressure at which the fluid becomes critical.

Figure 1 van der Waals equation of state.
The phase properties of a fluid are determined by intermolecular forces. In Figure 2, attractive forces are marked with a red color and repulsive forces with a green color. Attractive forces only act over short distances while repulsive forces act over both short and long distances. The attractive forces dominate in the liquid region, and the repulsive forces in the gas region.

Figure 2 Intermolecular attractive (red color) and repulsive (green color) forces.
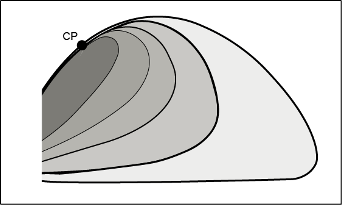
As shown in Figure 3, the gas and liquid properties approach each other when the critical point is approached. This means that the attractive and repulsive forces are also approaching each other. At the critical point, the gas and liquid phases are identical. At this point, there is a perfect balance between attractive and repulsive forces. van der Waals understood that an equation of state must reflect this balance, and this was what allowed him to develop a successful equation of state with the critical point as anchor point.

Figure 3 Gas and liquid properties of a pure hydrocarbon depending on the distance to the critical point (CP).
Today, the most widely used cubic equations of state are the SRK and PR equations. They are based on the same principles as the van der Waals equation, but to describe the development of vapor pressures of pure components with temperature more accurately, they use a temperature dependent a-parameter. Another important step forward is that these two equations are not limited to pure components, but can be applied to multi-component mixtures, including petroleum reservoir fluids. Figure 4 shows the SRK equation and its mixing rules.

Figure 4 Soave-Redlich-Kwong (SRK) equation for an N-component mixture.
Tc, Pc and are well-defined properties for inorganic gas components (N2, CO2 and H2S) and light hydrocarbons (C1-C6). For C7 and higher, PVT laboratories do not perform a complete component analysis but report boiling point fractions, where each fraction contains several components. Even with a complete component analysis, heavy hydrocarbons would present a challenge, as it is difficult to determine the critical point of a heavy hydrocarbon. It tends to decompose before reaching the temperature at which it would become critical.
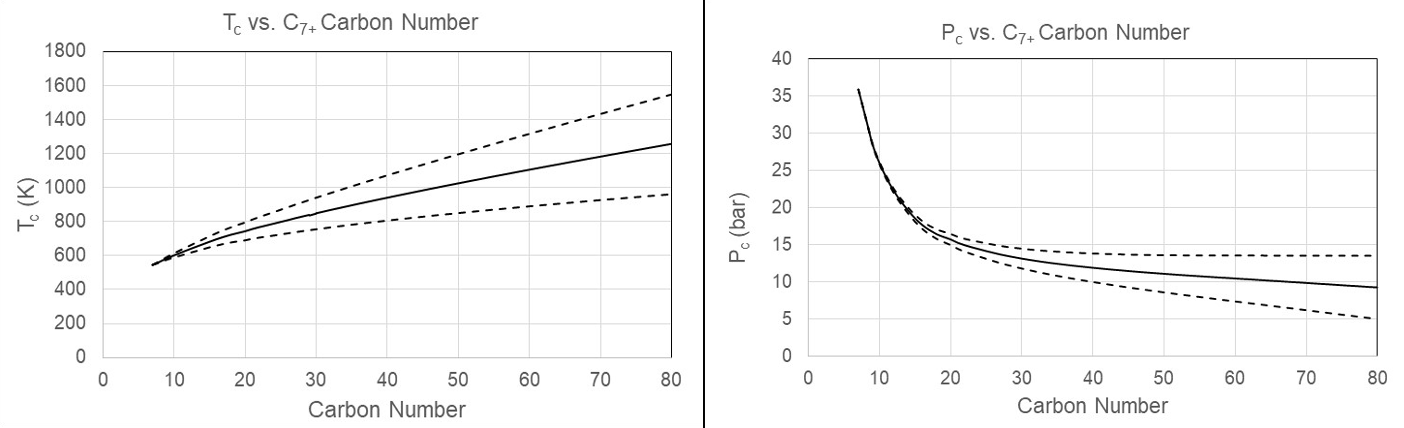
Instead, correlations are used for how Tc, Pc and of the C7+ fractions develop with carbon number. As shown in Figure 5, it is generally accepted that Tc should increase with molecular weight, Pc should decrease, and should increase to around C50, after which it may level off. The figure suggests upper and lower limits for Tc, Pc and , but no established rules exist for such limits.



Figure 5 Qualitative development of Tc, Pc and acentric factor of C7+ fractions with carbon number. The full drawn lines illustrate the default values, the dashed lines upper limits, and the dashed-dotted lines lower limits.
A recent SPE paper [1] suggests that the focus for C7+ fractions should be shifted from Tc, Pc and to the parameters a and b. The parameter a primarily affects the molecular attraction and b the repulsion. A correct representation of the phase behavior of a fluid requires the right balance between attraction and repulsion, which for C7+ components means a correct development of a/b with molecular weight.
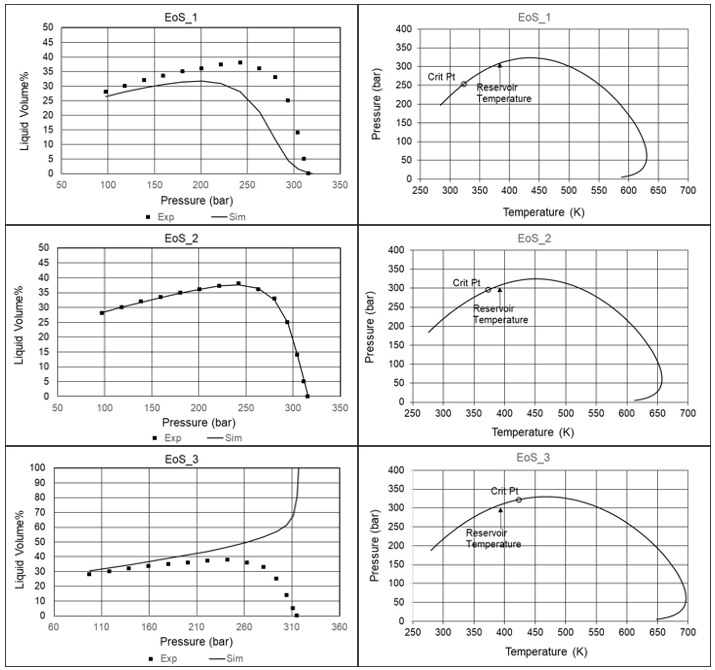
By using established correlations for the development of Tc, Pc and of the C7+ fractions with molecular weight, one will normally be assured against liquid-liquid split at temperatures above room temperature. However, one or more of the C7+ parameters will often have to be modified to get a better match to PVT data measured at the reservoir temperature. This is illustrated in Figure 6, where in one case Tc and in another case of the heaviest pseudo-component is increased relative to the value from the correlation. In both cases this introduces an unphysical 3-phase area, even though Tc and still develop monotonically with increasing molecular weight. An unphysical simulated 3-phase area is likely to give unphysical simulation results also at PT conditions other than in the simulated 3-phase area. With standard correlations, the fluid in question was correctly simulated to have a critical point, but the critical point is gone after the parameter adjustment.

Figure 6 Effect on the phase diagram of modifying Tc or of heaviest pseudo-component (red color) relative to the values from the standard correlations [2].
Figure 7 shows the developments of a/b and b for the individual C7+ pseudo-components when using standard correlations [2] for Tc, Pc and , and when Tc of the heaviest pseudo-component is modified as shown in Figure 6. With the standard correlations, approximately linear developments are seen for a/b versus molecular weight (M) and for b versus ln(M). When Tc of the heaviest component is modified, a/b and b for this component deviate from the linear relationships. The modification has destroyed the balance between attractive and repulsive intermolecular forces, and this is what has introduced an unphysical phase behavior. The mentioned recent SPE paper provides other examples showing that unphysical liquid-liquid splits can be avoided by ensuring that a/b of the C7+ pseudo-components develops linearly with molecular weight and that b develops linearly with the logarithm of molecular weight. These criteria can be used to quality check an EoS model which is not to simulate any liquid-liquid splits.

Figure 7 Ratio a/b and parameter b for the C7+ components plotted against molecular weight (M) and ln(M), respectively. The left-hand values are using standard correlations for Tc, Pc and [2]. The right-hand values are for Tc of the heaviest pseudo-component modified as shown in Figure 6.
Cases exist when an oil phase splits into two. As illustrated in Figure 8, a separate asphaltene liquid phase may precipitate from an oil phase when the pressure drops during production.

Figure 8 Asphaltene precipitation because of pressure drop.
Figure 9 shows plots of a/b and b for the C7+ components of a successful asphaltene simulation [3] using the SRK equation. For the non-asphaltene C7+ components, an approximate linear development is seen for a/b against molecular weight and for b against ln(molecular weight), while the asphaltene component stands out.


Figure 9 a(T)/b versus molecular weight (left) and b versus ln(molecular weight) (right) for the fluid compositions with asphaltenes
This example shows that the proposed analysis of how a/b and b should develop with molecular weight is not only a useful tool to quality check an EoS model that is not to simulate any liquid-liquid splits. It can also be used to force an EoS model to simulate an observed liquid-liquid split. In the latter case, a/b and b for the component dominating the second liquid phase must deviate from the linear trends seen for the other C7+ components.
References
[1] Pedersen, K.S., How to be in control of simulated liquid-liquid split when characterizing a reservoir fluid, SPE-231892-MS, SPE Europe Subsurface Conference, Bergen, Norway, April 21, 2026.
[2] Pedersen, K.S, Blilie, A.L. and Meisingset, K.K., PVT calculations on petroleum reservoir fluids using measured and estimated compositional data for the plus fraction, I&EC Research 31, 1378-1384, 1992.
[3] Pedersen, K.S., The mechanisms behind asphaltene precipitation – Successfully handled by a classical cubic equation, SPE-224534-MS, Gas and Oil Technology Showcase and Conference, Dubai, April 21-23, 2025.
Allocation of Feed Streams to Oil & Gas Process Plant
Figure 1 shows a schematic of a process plant that handles a mixture of three feed streams. The flow rates of each feed stream at elevated P & T are measured by a multiphase meter. Allocation calculations for such a flowsheet are often performed by summing the production that would be obtained if the input streams were processed individually. This is done either using black oil correlations or equation of state simulations. Such a procedure does not account for interactions between the feed streams in the process plant and will only give an approximate result for the total gas and oil production unless the inlet fluid compositions are (close to) identical.

Figure 1 Schematic of process plant
| Component | Fluid 1 Mol % |
Fluid 2 Mol% |
Fluid 3 Mol% |
|---|---|---|---|
| N2 | 0.528 | 0.477 | 0.560 |
| CO2 | 3.304 | 4.040 | 3.549 |
| C1 | 73.036 | 57.413 | 45.326 |
| C2 | 7.688 | 9.280 | 5.478 |
| C3 | 4.107 | 5.621 | 3.699 |
| iC4 | 0.700 | 1.004 | 0.700 |
| nC4 | 1.426 | 2.220 | 1.650 |
| iC5 | 0.538 | 0.831 | 0.730 |
| nC5 | 0.672 | 1.053 | 0.870 |
| C6 | 0.838 | 1.346 | 1.330 |
| C7 | 1.265 | 2.150 | 2.556 |
| C8 | 1.294 | 2.560 | 3.143 |
| C9 | 0.770 | 1.472 | 2.081 |
| C10+ | 3.835 | 10.532 | 28.329 |
| C7+ Molecular Weight | 169.5 | 195.5 | 254.9 |
| C7+ Density (g/cm3) | 0.8163 | 0.8315 | 0.8760 |
Table 1 Molar compositions of feed streams to the process plant.
| Component | Feed | Produced | ||||
|---|---|---|---|---|---|---|
| Fluid 1 mol/hr ×10-6 |
Fluid 2 mol/hr ×10-6 |
Fluid 3 mol/hr ×10-6 |
Mixture mol/hr ×10-6 |
STO Gas mol/hr ×10-6 |
STO Oil mol/hr ×10-6 |
|
| N2 | 0.401 | 0.220 | 0.261 | 0.882 | 0.882 | 0.000 |
| CO2 | 2.509 | 1.862 | 1.653 | 6.025 | 5.988 | 0.037 |
| C1 | 55.467 | 26.466 | 21.114 | 103.046 | 102.984 | 0.062 |
| C2 | 5.839 | 4.278 | 2.552 | 12.668 | 12.372 | 0.296 |
| C3 | 3.119 | 2.591 | 1.723 | 7.433 | 6.320 | 1.113 |
| iC4 | 0.532 | 0.463 | 0.326 | 1.320 | 0.868 | 0.452 |
| nC4 | 1.083 | 1.023 | 0.769 | 2.875 | 1.583 | 1.292 |
| iC5 | 0.409 | 0.383 | 0.340 | 1.132 | 0.369 | 0.763 |
| nC5 | 0.510 | 0.485 | 0.405 | 1.401 | 0.375 | 1.026 |
| C6 | 0.636 | 0.620 | 0.620 | 1.876 | 0.221 | 1.655 |
| C7-C13 | 3.812 | 4.665 | 7.342 | 15.819 | 0.079 | 15.740 |
| C14-C21 | 1.119 | 1.849 | 4.596 | 7.564 | 0.000 | 7.564 |
| C22-C32 | 0.406 | 0.863 | 2.934 | 4.203 | 0.000 | 4.203 |
| C33-C80 | 0.103 | 0.328 | 1.948 | 2.379 | 0.000 | 2.379 |
| Total | 75.945 | 46.097 | 46.582 | 168.624 | 132.042 | 36.582 |
Table 2 Molar component flow rates of feed streams and gas and oil product streams.
- Determination of the contributions of each feed stream to the molar component flow rates in the product streams.
- Conversion of the molar component flow rates in 1) to volumetric flow rates.
The feed stream numbers for C1 in Table 2 shows that the fraction of C1 molecules originating from feed stream 1 in the mixed feed is 55.467/(55.467+26.466+21.114) = 0.5383. This initial relationship is maintained throughout the process and the same applies to the other components. The first step in the allocation is therefore a material balance calculation for which the resulting molar component flow rates are shown in Table 3.
| Component | Produced Gas | Produced Oil | ||||
|---|---|---|---|---|---|---|
| Fluid 1 mol/hr ×10-6 |
Fluid 2 mol/hr ×10-6 |
Fluid 3 mol/hr ×10-6 |
Fluid 1 mol/hr ×10-6 |
Fluid 2 mol/hr ×10-6 |
Fluid 3 mol/hr ×10-6 |
|
| N2 | 0.401 | 0.220 | 0.261 | 0.000 | 0.000 | 0.000 |
| CO2 | 2.494 | 1.851 | 1.643 | 0.015 | 0.011 | 0.010 |
| C1 | 55.433 | 26.450 | 21.101 | 0.033 | 0.016 | 0.013 |
| C2 | 5.702 | 4.178 | 2.492 | 0.136 | 0.100 | 0.060 |
| C3 | 2.652 | 2.203 | 1.465 | 0.467 | 0.388 | 0.258 |
| iC4 | 0.350 | 0.304 | 0.214 | 0.182 | 0.158 | 0.112 |
| nC4 | 0.596 | 0.563 | 0.423 | 0.487 | 0.460 | 0.346 |
| iC5 | 0.133 | 0.125 | 0.111 | 0.276 | 0.258 | 0.229 |
| nC5 | 0.137 | 0.130 | 0.108 | 0.374 | 0.355 | 0.297 |
| C6 | 0.075 | 0.073 | 0.073 | 0.561 | 0.547 | 0.547 |
| C7-C13 | 0.019 | 0.023 | 0.037 | 3.793 | 4.642 | 7.305 |
| C14-C21 | 0.000 | 0.000 | 0.000 | 1.119 | 1.849 | 4.596 |
| C22-C32 | 0.000 | 0.000 | 0.000 | 0.406 | 0.863 | 2.934 |
| C33-C80 | 0.000 | 0.000 | 0.000 | 0.103 | 0.328 | 1.948 |
| Total | 59.469 | 36.097 | 36.476 | 16.476 | 10.000 | 10.106 |
Table 3 Molar component flow rates originating from each feed stream in the produced gas and oil.

Figure 2 How to derive the volumetric contributions of each feed steam to the product streams using partial molar volumes.
The partial molar volumes of each component in the produced gas and oil at standard conditions calculated using the volume corrected SRK equation are shown in Table 4.
| Partial Molar Volumes Produced Gas m3/mol |
Partial Molar Volumes Produced Oil m3/mol |
|
|---|---|---|
| N2 | 2.375×10-2 | 50.23 ×10-6 |
| CO2 | 2.362 ×10-2 | 47.94 ×10-6 |
| C1 | 2.368 ×10-2 | 52.03 ×10-6 |
| C2 | 2.354 ×10-2 | 64.07×10-6 |
| C3 | 2.343 ×10-2 | 79.15×10-6 |
| iC4 | 2.334 ×10-2 | 95.17 ×10-6 |
| nC4 | 2.332 ×10-2 | 94.06 ×10-6 |
| iC5 | 2.323 ×10-2 | 108.66 ×10-6 |
| nC5 | 2.320 ×10-2 | 109.45 ×10-6 |
| C6 | 2.308 ×10-2 | 126.96 ×10-6 |
| C7-C13 | 2.275 ×10-2 | 162.00 ×10-6 |
| C14-C21 | 2.201×10-2 | 276.09 ×10-6 |
| C22-C32 | 2.132 ×10-2 | 402.73 ×10-6 |
| C33-C80 | 2.037 ×10-2 | 630.69 ×10-6 |
Table 4 Partial molar volumes of each component in the produced gas and oil at standard conditions.
Using the partial molar volumes in Table 4 and the expression for the volumetric contribution of each feed stream in Figure 2, the allocation calculation can be completed. The volumetric contributions of each feed stream can be seen in Figure 1.
Compared to an allocation based on processing of individual feed streams, the proposed concept is more accurate when the feed streams have large variations in gas-oil ratio. It can be seen from Table 2 that almost all components lighter than C3 will end up in the produced gas, while components heavier than C6 will almost exclusively be found in the produced oil. The process simulation is therefore primarily to determine how the C3-C6 components distribute between gas and oil. A correct picture of this requires that the process simulation is performed on the mixed feed and not by combining data from processing of individual streams.
What Determines a Reservoir Fluid Composition?
Although the compositions of reservoir fluids vary, they exhibit certain common patterns including an approximately linear relationship between the logarithm of the mole percent of C10+ components and carbon number. This signals a non-random breakdown of the organic material that oil and gas originate from. A recent ADIPEC paper [1] concludes that a reservoir fluid that is allowed to age long enough will reach a state of chemical reaction equilibrium, but not all reservoir fluids have yet reached this point. An analysis of the compositions of several reservoir fluids from around the world led to the conclusion that the C7+ components are the first to reach reaction equilibrium, while the C1-C6 components are the last. By using data available in the early phase of an exploration process, it is possible to determine the stage of the degradation process and provide a good estimate of the current fluid composition. This allows both fluid properties and possible communication between different zones to be assessed at an early stage in the exploration of a reservoir. This note summarizes the ideas and conclusions presented in the ADIPEC paper.
Origin of Reservoir Fluids
As sketched in Figure 1, petroleum reservoir fluids are formed by decomposition of the remains of plants and animals, primarily marine organisms.

Figure 1 How oil and gas were formed.
Figure 2 illustrates that the organic material that led to the formation of oil and gas started with large molecules that were gradually broken down into the smaller molecules that make up gas and oil today. Molecules with 10 or more carbon atoms organized themselves in such a way that there is an approximately linear relationship between ln(mol%) and carbon number.

Figure 2 Decomposition process leading to a C10+ composition with a linear relationship between ln(mol%) and carbon number.
Chemical Reaction Equilibrium
A possible explanation for the observed systematics in the distribution of heavy components could be that reservoir fluids are not only in phase equilibrium but also in chemical reaction equilibrium. This would mean that the hydrogen and carbon atoms are combined in an energetically optimal way. If there was an excess of hydrogen (H) atoms relative to carbon (C) atoms, and the formation took place in a vacuum, the result would be pure methane, because methane has the lowest energy of formation.

Figure 3 Gibbs energies of formation for the lighter hydrocarbons showing that methane (C1) would be the favored component in vacuum.
However, Figure 3 does not give a correct picture. Methane requires 4 H-atoms per C-atom, which exceeds the H/C ratio in the organic source material. Also, the conversion into oil and gas does not take place at vacuum but at high pressure. As illustrated in Figure 4, an increasing C1 concentration means an increasing C1 fugacity, which will favor other hydrocarbon components.

Figure 4 Illustration of the fact that an increasing C1 concentration makes C1 less favorable.
The theory that is required to balance the contributions from formation energy, phase equilibrium and H/C ratio is outlined in a paper presented at ADIPEC 2025 [1]. With reservoir P & T and H/C ratio as input, the compositions that will be in chemical reaction equilibrium in the reservoir are calculated. Figure 5 shows four examples of a good agreement between a simulated equilibrium fluid composition and the actual reservoir fluid composition.

Figure 5 Examples of reservoir fluids at chemical reaction equilibrium at reservoir conditions. Detailed compositions can be found in the ADIPEC paper [1].
Non-Equilibrium Fluids
Not all reservoir fluids are in chemical reaction equilibrium. Figure 6 shows two reservoir fluid compositions that are not in equilibrium at reservoir conditions. For these two fluids as well as for several others covered by the analysis, the C1 concentration in the actual fluid is lower than in the equilibrium fluid, while the concentrations of C2-C6 are higher. For C7+, there is good agreement between the concentration in the reservoir fluid and in the simulated equilibrium fluid.

Figure 6 Examples of reservoir fluids not at chemical reaction equilibrium at reservoir conditions. Detailed compositions can be found in the ADIPEC paper [1].
The correspondence between the mole percent of C7+ in non-equilibrium reservoir fluids and equilibrium fluids is the key to understanding how the decomposition of organic material occurs. The high molecular weight compounds in the organic source material gradually break down into smaller molecules. The largest molecules are the ones to first reach equilibrium, while, as illustrated in Figure 7, the final step before equilibrium is reached, is the breakdown of C2-C6 molecules into smaller molecules, including C1.

Figure 7 Final step towards chemical reaction equilibrium.
Reservoir Fluid Compositions from Early-Stage Exploration Data
The density of a reservoir fluid may be available in the early exploration phase, which together with reservoir P and T enables a good estimate of the mole percent of C1. A non-equilibrium fluid will have a lower C1 mole percent than an equilibrium fluid. Distributing the extra amount of C1 in the equilibrium fluid between the C2-C6 components, as shown in Figure 8, gives a good estimate of the current reservoir fluid.

Figure 8 ”Repair” technique applied to a composition at chemical reaction equilibrium to obtain the actual reservoir fluid composition.
Case Study
Figure 9 is a sketch of a North Sea reservoir with four separate formations at approximately the same reservoir conditions. Although there is no communication between the individual formations, the fluid compositions in three of the four formations (A-C) are almost identical while the composition of the fluid in the last formation (D) is different.

Figure 9 Sketch of North Sea reservoir with 4 separate formations.
In Formation A, the fluid column extends over a vertical zone of more than 50 meters and there is a slight compositional gradient. As can be seen from Figure 10, the equilibrium fluid represents an approximate average of the fluid composition in the column. In Formation B, two nearly identical fluid compositions matching the equilibrium composition were sampled at a vertical distance of less than 10 meters. In Formation C, two samples were taken more than 40 meters apart, which is a sufficient vertical distance to expect some compositional grading, but none is seen. Both samples correspond to the simulated equilibrium composition, suggesting that Formation C may in fact be two separate formations. The fluid in Formation D is not at chemical reaction equilibrium but shows the same kind of deviations from an equilibrium fluid as is seen for the two non-equilibrium fluids in Figure 6. It is therefore a suitable candidate for the “repair” technique outlined in Figure 8.

Figure 10 Comparison of observed and chemical reaction equilibrium fluids for North Sea reservoir with 4 separate formations.
The “repair” carried out for the reservoir fluid in Formation D is sketched in Figure 11 and resulted in a fluid composition accurately matching the observed fluid in Formation D.

Figure 11 “Repair” of equilibrium composition in Formation D of North Sea Reservoir.
Applications
Not all reservoir fluids are at chemical reaction equilibrium at reservoir conditions, but even for such fluids it is possible to predict the composition of a reservoir fluid solely from reservoir P and T and in situ density. This enables simulations that require a fluid composition to be started at an early stage of exploration. When similar fluid compositions are found in neighboring formations, it will often be seen as a sign of communication between the formations. As shown in the above example, the reason may instead be that the fluids are at chemical reaction equilibrium, and this may help rule out any doubt about fluid communication. How much a fluid composition deviates from chemical reaction equilibrium provides a measure of the stage of chemical degradation, a knowledge that can also be useful in geochemical studies.
References
Pedersen, K.S., Hjermstad H.P., Diesen, M., Reservoir Fluid Compositions – A Result of Chemical Reaction Equilibrium and Gravity Segregation, SPE-229909-MS, ADIPEC November 2-5, 2025.
When to Use Non-Zero Binary Interaction Parameters in Equation of State Modeling
Cubic equations of state were developed for pure components with the critical point as a fixed point. This fixed point no longer exists when dealing with mixtures, and the reliability of equation of state simulations for mixtures depends on the suitability of the mixing rules. SRK and PR are the most widely used cubic equations. These are 2-constant equations, where the two constants are a and b. While b is a volume parameter, a is a key parameter in the attractive term. When Soave proposed the currently used mixing rules, he introduced a correction term in the mixing rule for the a-parameter, (1-kij), where kij is a binary interaction parameter that by default is zero but can be assigned a non-zero value when the mixing rule with kij=0 is inaccurate. However, non-zero kij‘s can cause harm if used incorrectly.
SRK Equation
The Soave-Redlich-Kwong (SRK) equation and the mixing rules proposed by Soave, and also used with the PR equation, are shown in Figure 1. The equation consists of two terms, a repulsive term, derived from the ideal gas equation, and an attractive term, (a/(V(V-b))). For a mixture, the a-parameter includes the square root of the product of the a-parameters for each pair of individual components. For pairs of different components, it is possible to correct this term by a factor (1-kij) and assign kij a non-zero value, while kij is zero for pairs of identical components. A positive kij will reduce the a-parameter of the mixture and increase the mixture molar volume, which leads to a reduction of the attractive term of the equation of state.

Figure 1 Soave-Redlich-Kwong (SRK) equation [1].
Figure 2 illustrates the influence of a binary interaction parameter. A spring holds two spheres together. For kij = 0 it is a tight spring, while it is looser for kij > 0. A kij > 0 means less attraction and a higher molar volume (lower density).

Figure 2 Illustration of impact of the binary interaction parameter. kij = 0 corresponds to a tight spring, while the spring is looser for kij > 0.
Equation of state modeling for petroleum reservoir fluids distinguishes between defined components and pseudo-components. Defined components are those with well-defined critical properties, while pseudo-components represent a range of components with comparable properties and do not have well-defined critical properties.
Mixtures of Defined Hydrocarbons
Consider a mixture of equal molar amounts of C1, nC5, and nC10. Figure 3 shows simulated phase diagrams for this mixture with all binary interaction parameters equal to zero (left) and with the binary interaction parameter for nC5-nC10 set to 0.15 (right). For temperatures above 60°C, the two diagrams are almost identical, but at lower temperatures, the mixture with a kij = 0.15 for nC5-nC10 has a 3-phase area consisting of one vapor phase and two liquid phases.


Figure 3 Simulated phase diagrams for equimolar mixture of C1, nC5, and nC10 using the SRK equation. V stands for vapor and L for liquid.
Table 1 shows two sets of multiphase PT flash results for the equimolar mixture of C1, nC5, and nC10 at 20 bar and 50°C, one for all kij‘s equal to zero, and one for the binary interaction parameter for nC5-nC10 equal to 0.15. The non-zero kij causes a liquid-liquid split, with one liquid phase dominated by nC5 and the other one by nC10. The positive kij has, as illustrated in Figure 2, reduced the simulated attraction between nC5 and nC10, and this leads to an unphysical liquid-liquid split. Hydrocarbons are non-polar compounds whose molecules do not carry any positive or negative charges. For such compounds, it is inappropriate to correct the a-parameter using non-zero binary interaction parameters.

Table 1 Multiphase PT flash results with SRK equation for equimolar mixture of C1, nC5, and nC10 at 20 bar and 50°C. The shown liquid-liquid split for kij = 0.15 is unphysical and an example of incorrect use of a non-zero binary interaction parameter.
Mixtures with CO2
CO2 consists of elongated molecules that, in liquid form, orient themselves in parallel, which favors a high density, as illustrated in Figure 4.

Figure 4 Phase diagram for CO2, illustrating the influence of the elongated molecular shape on the phase densities.
CO2 and propane (C3) have almost the same molecular weight, but their vapor pressure curves are quite different as can be seen in Figure 5. CO2 cannot maintain its ordered liquid structure at temperatures above 31°C, which is the reason for its low critical temperature. Because the vapor phase is less structured than the liquid, it requires a high pressure (73.76 bar) for CO2 to reach the critical point where two identical phases are in equilibrium. Propane forms a less dense and less ordered liquid phase than CO2, and can maintain the liquid state until the temperature is above 95°C. The less ordered structure also means less difference in how the propane molecules arrange themselves in the gas and liquid states, and propane reaches the critical pressure with two identical phases in equilibrium at a significantly lower pressure than CO2.

Figure 5 Vapor pressure curves of CO2 and propane (C3).
When mixing CO2 with propane, CO2 cannot maintain the ordered liquid structure it had in pure form. The distance between the CO2 molecules increases. This can be modeled by assigning the component pair CO2-C3 a kij of 0.12. As shown in Figure 6, this gives rise to a modest reduction in the a-parameter (left), which leads to a significant increase in the molar volume (right), corresponding to the spring in Figure 2 being loosened.


Figure 6 Influence of kij on the a-parameter for CO2-C3 mixtures at 21.1°C (left), and on the molar volume at 21.1°C and 60 bar (right). The volume calculations were made with the volume corrected SRK equation.
Figure 7 shows an almost perfect match of measured dew and bubble point pressures for mixtures of CO2 and C3 at 21.1°C with kij = 0.12, while significant deviations are seen for kij = 0. The kij of 0.12 for CO2-C3 is an example of correct use of a non-zero binary interaction parameter.

Figure 7 Experimental [2] and simulated dew and bubble point pressures of CO2-C3 mixtures at 21.1°C.
Mixtures with Water (H2O)
Water (H2O) forms an even denser and more structured liquid phase than CO2 and a non-zero kij is not sufficient to match the mutual solubility of water and a hydrocarbon. It requires a mixing rule that accounts for the special structure of water. However, for many engineering applications it is sufficient to know the water concentration in a hydrocarbon phase in contact with water, while it is acceptable to consider the water phase as pure water. When this is the case, a non-zero kij can be used. Table 2 shows that the measured solubility of H2O in nC4 at 38°C and 200 bar is 0.1 mol% [3], which simulation result is obtained with kij = 0.426. With kij = 0 the simulated water concentration in the hydrocarbon phase is more than 20 times higher. This use of a non-zero binary interaction parameter for a mixture of water and a hydrocarbon is pushing the limits of what a classical cubic equation of state can manage, but it is sufficient for many engineering purposes.

Table 2 Experimental [3] and simulated solubilities of H2O in nC4 at 200 bar and 38°C.
C7+ Pseudo-Components
Petroleum reservoir fluids contain thousands of hydrocarbon components with 7 or more carbon atoms, for which the critical properties are mostly unknown. To perform equation of state calculations for reservoir fluids, the C7+ components must be grouped to an appropriate number of pseudo-components, and each pseudo-component assigned a critical temperature, a critical pressure, and an acentric factor, and each component pair a binary interaction parameter.
Table 3 shows a characterized reservoir oil composition. Two sets of critical temperatures are shown for the three C7+ pseudo-components. The critical temperatures shown in black are the standard values, while those in red are slightly modified. Similarly, the binary interaction coefficients for C1-C7+ can either assume the standard value of zero or the value of 0.04 shown in red in Table 4. The full drawn red line in Figure 8 shows the phase envelope simulated with the critical temperatures marked in red in Table 3 and kij values of zero for C1-C7+. This gives a simulated saturation point at the reservoir temperature of 97.5°C that is 8.6% lower than the experimentally determined saturation pressure of 200 bar.

Table 3 Reservoir fluid composition characterized for the volume corrected SRK equation. The red C7+ Tc‘s are non-standard values.

Table 4 Non-zero binary interaction parameters for use with the fluid composition in Table 3. The red kij‘s are non-standard values.
A non-zero kij for C1-C7+ will reduce the attraction between these components. It will make C1 more volatile, and a gas phase will form at a higher pressure than for kij = 0. Therefore, it may be tempting to use non-zero kij‘s to eliminate small deviations between the simulated and measured saturation pressures, and Figure 8 shows that the correct saturation pressure can be simulated by assigning the kij‘s for C1-C7+ the red values of 0.04 in Table 4.

Figure 8 Experimental saturation point and simulated phase envelopes for the fluid in Tables 3 and 4. The red Tc‘s in Table 3 are used.
A differential liberation experiment [4] has been conducted for the oil composition in Table 3. Figure 9 shows experimental and simulated oil formation volume factors (Bo) and solution gas/oil ratios (Rsd). The simulation results are for the red (non-standard) parameters in Tables 3 and 4, which implies a kij of 0.04 for C1-C7+. The simulated oil formation factors and gas oil ratios are seen to be too high.


Figure 9 Experimental [4] and simulated differential liberation results the fluid in Tables 3 and 4. The red Tc‘s in Table 3 are used.
This is an example of an incorrect use of non-zero binary interaction parameters. The simulated saturation pressure for kij = 0 in Figure 5 is too low because the Tc‘s of the C7+ components are too low and not because the attraction between C1 and C7+ is too high. As can be seen from Figure 1, lower Tc values mean lower a-parameters and thus a lower overall molecular attraction. Too large a proportion of the C7+ pseudo-components, and especially of C7-C14, end up in the gas phase at low pressure, causing the simulated Bo’s and Rsd’s to be too high as seen in Figure 9. The cure is not to use non-zero binary interaction parameters but to increase the Tc‘s of the C7+-components to the values shown in black in Table 3. These values give an almost perfect match of the measured Bo’s and Rsd’s as seen in Figure 10.


Figure 10 Experimental [4] and simulated differential liberation results the fluid in Tables 3 and 4. The black Tc‘s in Table 3 are used.
Asphaltenes
Asphaltenes differ from other hydrocarbons in being almost insoluble in light n-paraffins. This can be modeled by assigning component pairs consisting of one gas and one asphaltene component a kij between 0.01 and 0.06 [5]. Figure 11 shows an example of an asphaltene phase diagram. A positive kij for pairs of a gas and an asphaltene component reduces the attraction between the two component groups and leads to the formation of a separate liquid phase dominated by asphaltenes. At pressures higher than the bubble point pressure, the asphaltene phase coexists with an oil phase. This is an example of a non-zero kij used to model an observed liquid-liquid split.

Figure 11 Simulated asphaltene phase diagram with measured data for asphaltene onset pressures and bubble point pressures [5].
Summary
All parameters involved in the calculation of the parameters a and b in a cubic equation of state have a physical meaning. This also applies to the binary interaction parameters. In pure form, CO2, water, and asphaltenes form dense structures that change to looser structures when mixed with other components. Non-zero binary interaction parameters are designed to handle this phenomenon, and the use of non-zero binary interaction parameters should be reserved for such cases and not used to try to correct for unfortunate choices of Tc‘s or Pc‘s of C7+ pseudo-components.
References
[1] Soave, G., “Equilibrium Constants from a Modified Redlich-Kwong Equation of State”. Chem. Eng. Sci. 27, 1197-1203, 1972.
[2] Reamer, H.H., Sage, B.H. and Lacey, W.N., “Phase Equilibria in Hydrocarbon Systems. Volumetric and Phase Behavior of the
Propane-Carbon Dioxide System”, Ind. Eng. Chem. 43, 2515–2520, 1951.
[3] Reamer, H.H., Sage, B.H. and Lacey, W.N., Chem., “Phase Equilibria in Hydrocarbon Systems. n-Butane-Water System in the Two-
Phase Region”, Ind. Eng. Chem. 44, 609–615, 1952.
[4] Pedersen, K. S., Christensen, P.L. and Shaikh, J.A., Phase Behavior of Petroleum Reservoir Fluids, 3rd edition, Taylor & Francis, Boca Raton, US, 2024.
[5] Pedersen, K. S., “The Mechanisms Behind Asphaltene Precipitation – Successfully Handled by a Classical Cubic Equation”,
SPE-224534-MS, Gas and Oil Technology Showcase and Conference, Dubai, April 21-23, 2025.
150 Years of History of Cubic Equations of State
After van der Waals presented the first cubic equation of state in 1873, it took more than 100 years for cubic equations to become the invaluable tool in the petroleum industry they are today. This had several reasons, one being that the early petroleum industry did not need such a tool. The need arose when the production of gas and oil from deep reservoirs at high pressures and temperatures took off in the 1970s, but there were still obstacles to be overcome. New algorithms had to be developed to calculate phase amounts and phase compositions throughout the PT region, including the near-critical region. Since cubic equations were developed for hydrocarbon mixtures, they were not in their original form applicable to mixtures with water and other polar components. The gap between the equations of state and the models used in the chemical industry for mixtures with polar components had to be closed. This technical note tells the story of cubic equations and what has made a 150-year-old model concept survive and remain unsurpassed.
In 1873, Johannes Diderik van der Waals defended his doctoral thesis entitled: “On the Continuity of the Liquid and Gaseous States” [1], in which he argued that the properties of a pure component in both the gaseous and liquid states are determined by the location of the critical point. This was only four years after the existence of a critical point had been documented [2] and at a time when it was not yet fully accepted that fluids are made up of molecules. Scientists still considered the gas and liquid states to be independent. It must have required an eminent physical understanding and a certain degree of courage to put forward a theory that there is a continuity between the gas and liquid states. As illustrated in Figure 1, van der Waals claimed that gas and liquid coexist at the vapor pressure thanks to a balance between repulsive and attractive molecular forces. The gas phase is dominated by repulsive forces, and the liquid phase by attractive forces. Moving up the vapor pressure curve towards the critical point, the molecular forces in the two phases gradually become more similar, and at the critical point the two phases are identical. At pressures and temperatures higher than at the critical point, a phase separation can no longer occur.

Figure 1 How van der Waals saw the gas and liquid densities develop with temperature.
van der Waals expressed his findings in the equation shown in Figure 2. It says that the pressure is determined by two opposing molecular interactions, one repulsive and one attractive. The repulsive term is a slight modification of the ideal gas equation, while the attractive term expresses that two molecules will attract each other with a force that increases with the inverse of the squared molar volume, V.

Figure 2 Van der Waals equation of state [1].
Figure 3 shows qualitative PV curves at three different temperatures. At temperatures, T, higher than the critical temperature, Tc, the PV curve has a hyperbolic-like shape known from the ideal gas equation. At Tc the PV curve has what van der Waals interpreted as an inflexion point at the critical pressure, Pc. This made it possible to express the parameters a and b as unique functions of Tc and Pc, thus expressing van der Waals’ main point, that the properties of a pure component in the entire PT region are determined by the location of the critical point. At temperatures lower than Tc, the PV curve has a horizontal section where a reduction in volume does not change the pressure. On this horizontal section, gas and liquid coexist, and a phase transition from gas to liquid takes place. The van der Waals (vdW) equation cannot represent the horizontal part of the phase diagram (dashed in Figure 3), but it represents the endpoints and thus the molar volumes of the gas and liquid phases present during the phase transition. The vdW equation can be rewritten as a third-degree equation in molar volume and is therefore also called the first cubic equation of state. In 1910 van der Waals received the Nobel prize for his work on this equation.

Figure 3: PV isotherms of a pure component at three different temperatures.
For many years, the vdW equation was more of an academic discovery than a practical tool. The application was limited to pure components, and the accuracy was not considered sufficient for use in engineering calculations. In 1949, Redlich and Kwong (RK) [3] presented a slightly modified cubic equation of state. They were the first to suggest how to extend the use of a cubic equation of state to multi-component mixtures. The different molecules were represented as one average molecule as sketched in Figure 4. This was a major step forward and the RK equation is considered the first modern cubic equation of state.

Figure 4 The essence of the mixing rules for cubic equations of state.
Today, the Soave modification (SRK) from 1972 [4] is more widely used than the RK equation. Soave found that the variation of the vapor pressure with temperature was more curved than predicted by the vdW and RK equations. A few years earlier, Pitzer had introduced the acentric factor [5] as a measure of the curvature of a vapor pressure curve relative to that of argon. Soave modified the curvature of the vdW vapor pressure curve by making the a-parameter a function of a 2nd order polynomial in the acentric factor. Soave’s equation is shown in Figure 5.

Figure 5 Soave-Redlich-Kwong (SRK) equation [4].
Figure 6 illustrates Soave’s correction to the vapor pressure curve that makes use of the acentric factor.

Figure 6 Soave’s modification to the vdW vapor pressure curve [4].
Inorganic components like N2, CO2 and H2S often appear in petroleum reservoir fluids. These components have strong mutual attractive forces, while the interaction is less between an inorganic component and a hydrocarbon. To better describe the interaction between inorganics and hydrocarbons, Soave introduced a correction to the a-parameter called a binary interaction parameter, kij. For a component pair consisting of a hydrocarbon and an inorganic component, kij is typically in the range 0.04-0.15.
Soave’s equation predicted too low liquid densities. In 1976 Peng and Robinson (PR) proposed a modified cubic equation of state [6] that reduced the discrepancy seen with the SRK equation between experimental and calculated liquid densities, but it was not until 1982 satisfactory results for liquid densities were obtained with a cubic equation of state. As illustrated in Figure 7, Peneloux [7] obtained accurate liquid densities by horizontally shifting the PV curves by a distance, c, to have them match experimental liquid molar volumes.

Figure 7 Volume translation concept by Peneloux et al. [7].
Figure 8 shows how the SRK equation and the Peneloux (Pen) volume-corrected equation are related.

Figure 8 Relation between SRK equation [4] and Peneloux volume corrected equation [7].
Accurate phase densities are essential, but the most important application of an equation of state is to calculate the amounts and compositions of two or more phases in equilibrium. At equilibrium, all components in each phase will have the same fugacity, f. The left-hand container in Figure 9 shows a system that is not in equilibrium. All components in the gas phase have a higher fugacity than the same components in the liquid phase, and there is a net transport of components from the gas phase to the liquid phase. Fugacity can be seen as a measure of the tendency to escape. In the right-hand container, all components have the same fugacity in both the gas and liquid phases, and there is no net transport across the phase boundary. The fluid system is in equilibrium.

Figure 9 Non-equilibrium system (left-hand) and equilibrium system (right-hand).
Carl von Linde, the founder of the Linde chemical company, became familiar with the van der Waals equation as early as 1871/72, which was before van der Waals published the equation, and he tried to use the equation to solve multi-component phase equilibrium problems [8]. He had to give up because the calculations were so cumbersome that he found it almost impossible to solve them manually. The availability of computer power was one of the reasons the use of cubic equations of state took off in the late 1970s.
Even after computers became available, it was unclear how to determine whether a fluid system would remain single-phase or split into two or more phases. Gibbs had already described a procedure for this around 1900 [9]. Among the possible states, a fluid system will choose the one with the lowest Gibbs energy. However, Gibbs did not have the computational tools needed to make full use of his discovery, and it went almost unnoticed until Professor Michelsen in the early 1980s became aware of it and used it to develop efficient computer algorithms [10].
Water is often produced together with oil and gas. Some of the water may have been dissolved in the hydrocarbon phases in the reservoir, and some may come from an underlying formation water zone. Cubic equations were long considered unsuitable for handling mixtures with water because water is a polar component with strong mutual attractive forces. As shown in Figure 10, water molecules dissolved in a hydrocarbon (HC) phase will not be evenly distributed but stick together and form H2O-rich pockets. The figure also shows that something similar is seen for hydrocarbons dissolved in a water phase. The classical mixing rules shown in Figure 5, assume that all molecules are distributed evenly in both gas and liquid phases.

Figure 10 Close-up of molecules in a hydrocarbon phase saturated with water and in a water phase saturated with hydrocarbons. Mixing rule of Huron and Vidal for the a-parameter.
At the time cubic equations became frequently used in the oil industry, the chemical industry had already for several years successfully been using so-called GE models that could handle mixtures with polar components. These models describe what difference it makes to the interaction between two molecules whether they are identical or not. The GE models had the drawback that they were limited to liquid phases and low pressure. They were therefore not immediately applicable in the petroleum industry.
Models were proposed that incorporated the qualities of a GE model into a cubic equation of state, but most of them had the disadvantage that they required interaction parameters to be determined for all pairs of dissimilar molecules, and it could not be guaranteed that a calculation with negligible amounts of water would converge towards the result that would be obtained for a water free mixture using classical mixing rules. A publication by Huron and Vidal [11] stood out. The attractive term, a, had been rewritten to be a function of GE at infinite pressure (see Figure 10) in such a way that new interaction parameters were only required for component pairs with water or another polar component.
This is where the story of cubic equations of state ends for now. Cubic equations of state have undergone a great deal of development since 1873, so one may wonder why the van der Waals equation continues to be referred to. This is because van der Waals was the first to realize that the location of the critical point of a fluid exerts a decisive influence on the phase behavior, not only near the critical point, but throughout the gas and liquid regions. None of the improvements to the original equation described above have challenged this assumption.
References
- van der Waals, J.D., “O ver de Continuiteit var der Gas- en Vloeistoftoestand”, Doctoral Dissertation, Leiden University, The Netherlands, 1873 (In Dutch).
- Andrews, T. “On the Continuity of the Gaseous and Liquid States of Matter”, Philosophical Transactions of the Royal Society of London, vol. 159, 575–590, 1869.
- Redlich, O. and Kwong, J.N.S., “On the Thermodynamics of Solutions. V. An Equation of State. Fugacities of Gaseous Solutions”, Chem Review 44, 233-244, 1949.
- Soave, G., “Equilibrium Constants from a Modified Redlich-Kwong Equation of State”. Chem. Eng. Sci. 27, 1197-1203, 1972.
- Pitzer, K., “Volumetric and Thermodynamic Properties of Fluids. I. Theoretical Basis and Virial Coefficients”, J. Am. Chem. Soc. 77, 3427-3433, 1955.
- Peng, D,-Y. and Robinson, D.B., “A New Two-Constant Equation of State”, Ind. Eng. Chem. Fundam. 15, 59-64,1976.
- Péneloux, A., Rauzy, E. and Fréze, R., “A Consistent Correction for Redlich-Kwong-Soave Volumes”, Fluid Phase Equilibria 8, 7-23, 1982.
- Burr, P. et al. “Professor Michelsen’s Impact on Physical Property Prediction at Linde Engineering and Ideas for Future Directions”, Fluid Phase Equilibria 580, 114019, 2024.
- “The Collected Works of J. Willard Gibbs”, Nature 124, 119–120, 1929.
- Michelsen, M.L., “The Isothermal Flash Problem, Part I. Stability. Part II. Phase-Split Calculation”, Fluid Phase Equilibria 9, 1-41, 1982.
- Huron, M.J. and Vidal, J., “New Mixing Rule in Simple Equations of State for Representing Vapor-Liquid Equilibria of Strongly Non- Ideal Mixtures”, Fluid Phase Equilibria 3, 255-271, 1979.
What Causes Asphaltenes to Precipitate and How is it Modeled?
Figure 1 shows four examples of phase diagrams for oils containing asphaltenes. For Type A and B reservoir fluids, the upper asphaltene onset pressure (AOP) decreases with increasing temperature in the temperature range relevant to reservoir fluid production. Type A fluids will precipitate asphaltenes in the reservoir if the pressure reaches the upper AOP. Type B fluids have a temperature range in which asphaltene precipitation does not occur regardless of pressure. If the reservoir temperature is in this range, asphaltene precipitation will not occur in the reservoir but may occur in the well or in the process plant where the temperature is lower. For both Type A and B fluids, the slope of the upper AOP curve changes to increase with temperature at higher temperature. This shift usually occurs at a temperature that is too high to be relevant for oil production, but it might be relevant under refinery conditions. The Type C phase diagram is similar to the Type B diagram but lacks the asphaltene precipitation region at elevated temperature. In rare cases, the upper AOP is seen to increase with temperature at typical reservoir and production temperatures. This is illustrated by the Type D phase diagram in Figure 1. For all four types of phase diagrams, asphaltene precipitation occurs only in a limited pressure range. The highest amount of asphaltene precipitation is seen at the saturation pressure. Below the saturation pressure, evaporation of gas will reduce the amount of precipitated asphaltenes and eventually all asphaltenes will be dissolved in the oil phase. The pressure at which this occurs is referred to as the lower asphaltene onset pressure.





dG=VdP-SdT
G is Gibbs free energy, V is molar volume, S is entropy, and T is temperature. A change from a single-phase fluid to a two-phase fluid will occur if the Gibbs free energy of the fluid when split into two phases is lower than the Gibbs free energy, the fluid would have as a single-phase. At a fixed temperature, the last term in the above relation cancels out and the optimum state after a pressure reduction (negative dP) is the one (single-phase or two-phase) of the higher molar volume. This means that the asphaltene onset pressure is determined by the system parameters that are most influential on the total molar volume.
Figure 3 illustrates the two options that exist for an asphaltene-containing oil when the pressure drops. It can remain single-phase, or it can split into two phases, with the incipient phase being dominated by asphaltenes. The state of the higher volume is the stable one.

For any reservoir fluid, reducing the pressure at a constant temperature will make the volume increase, and so will the spacing of the molecules that make up the fluid. As illustrated in Figure 4, asphaltene precipitation will occur when the distance between the oil molecules becomes too large to keep the asphaltene molecules in solution.

One may ask whether this mechanism is consistent with the fact that resins are seen to play an important role in asphaltene precipitation. Resins are aromatic compounds and have a higher density (smaller intermolecular distance) than other hydrocarbon groups (paraffins and naphthenes), and it is primarily this property that makes reservoir fluids rich in resins less likely to precipitate asphaltenes.
For the oil industry, it is desirable to use the classical cubic equations of state for as many purposes as possible, as these equations are the industry standard. A recent paper [1] has shown that to date, no asphaltene data has been published that could not be satisfactorily modeled with a classical cubic equation of state. There is no need for more complex equations, such as CPA or PC-SAFT. Figure 5 shows examples of experimental asphaltene onset pressure data for Type A-D fluids along with simulation results with the SRK equation. Data sources and model parameters can be seen from the paper or obtained by contacting Kapexy Aps.




- [1] Pedersen, K. S., “The Mechanisms Behind Asphaltene Precipitation – Successfully Handled by a Classical Cubic Equation”, SPE-224534-MS, Gas and Oil Technology Showcase and Conference, Dubai , April 21-23, 2025.
Stages of Carbon Capture Storage
CO2 has several industrial uses, including supercritical extraction, fire extinguishing, refrigeration, and welding. Thanks to these many applications, data and validated models for CO2 are freely available on the Internet. In that light, one may wonder why Carbon Capture Storage (CCS) has developed into a new technical discipline that engages many engineers worldwide. It has because CCS involves transport and storage of CO2 quantities that are out of proportion to the amount of CO2 seen in industrial applications and takes place under other conditions. The weight of CO2 produced by burning fossil fuels is three times the weight of the fossil fuels being burned and amounted to 37.4 Gt in 2023. Over the years, the oil industry has built up reliable simulators for the production, transport and processing of large quantities of gas and oil, as well as for injecting gas into depleted oil fields. It seems natural to use the same models and methods to simulate CO2 transport and underground storage. However, CO2 differs from oil or gas and presents challenges not seen for hydrocarbon mixtures. This note reviews some of the characteristics of CO2 and some of the challenges that arise with CCS, but it starts with a historical review of the CO2 concentration in the atmosphere.
CO2 concentration in the atmosphere
Seen over a period of millions of years CO2 has an interesting and life-giving history. Without CO2, there would not have been the kind of life on earth we see today. The atmosphere today consists of about 78.08% nitrogen (N2), 20.95% oxygen (O2), 0.93% argon (Ar) and 0.04% (about 400 ppm) CO2, but it has not always been like that. The atmosphere would have contained no O2, had it not been formed by the photosynthesis of green plants, and without that more than 21% (210,000 ppm) of atmospheric air would have been CO2, disregarding the water content. From the day green plants arose around 700 million years ago, they began to consume CO2, which as shown in Figure 1 was used as a building material to form glucose (C6H12O6) and oxygen (O2). This process is called photosynthesis. For plants, O2 is a waste product, but the animals and humans who later populated the earth reversed the chemical reaction in Figure 1 by consuming plants and other animals. This released CO2, which in turn could be used as building material by the plants.
Figure 1 Photosynthesis.
Not all organisms are consumed by other organisms. Some are buried underground and over time turned into gas, oil, or coal. With each organism buried, humans and animals were giving less CO2 back to the atmosphere than consumed. A hundred years ago, the CO2 concentration in the atmosphere had dropped to about 280 ppm (0.028%). A drop in atmospheric CO2 concentration from more than 210,000 ppm when green plants appeared to around 280 ppm may seem quite dramatic but given that it happened over a period of about 700 million years, the annual decrease in CO2 concentration is only of the order of 0.3 parts per billion and has hardly been felt within a few generations. With a continued steady annual decline in CO2 concentration, it would take about a million years for the CO2 to be used up and all life would die out.
Throughout history, there have been fluctuations in the CO2 concentration in the atmosphere. Measurements suggest that the CO2 concentration during the last glacial period, about 20,000 years ago, was as low as 180 ppm. Cold water can dissolve more CO2 than hot water. Degassing of CO2 from seawater is therefore believed to be a major source of the increase in CO2 concentration in the atmosphere from about 180 ppm to 280 ppm that happened from the time about 30% of the earth was covered by ice to the start of the modern industrial age about 100 years ago.
CO2 is produced when burning methane (CH4), other hydrocarbons, and coal. This could be seen to bring some of the CO2 originally in the atmosphere back to the atmosphere. However, if more CO2 is produced from respiration of living organisms and by burning fossil fuels than consumed by plants and trees, the CO2 concentration in the atmosphere will increase as opposed to what has happened for the millions of years with a net removal of CO2 from the atmosphere to carbon compounds buried underground. If all the carbon were taken up from the underground and burned, one must expect the CO2 concentration to return to the 21% level it had when the first green plants appeared, in which environment no animal species would survive.
CO2 quantities produced by combustion
The chemical reaction for burning of methane (CH4) is:
CH4 + 2O2 → CO2 + 2H2O
Table 1 shows a typical reservoir oil composition. As is detailed in the table, burning 1 mole (=107.96 g) of oil will produce 344.18 g CO2. The weight amount of CO2 produced is in other words 344.18/107.96 = 3.19 times higher than the weight amount of the reservoir oil.
| Oil
component |
Mol%
of oil |
Molecular
weight |
Grams oil constituent
per mole oil |
Grams carbon
per mol oil |
Grams CO2
produced per mole oil |
| N2 | 0.56 | 28.01 | 0.16 | 0.00 | 0.00 |
| CO2 | 3.55 | 44.01 | 1.56 | 0.43 | 1.56 |
| C1 | 45.33 | 16.04 | 7.27 | 5.44 | 19.94 |
| C2 | 5.48 | 30.07 | 1.65 | 1.31 | 4.82 |
| C3 | 3.70 | 44.10 | 1.63 | 1.33 | 4.88 |
| iC4 | 0.70 | 58.12 | 0.41 | 0.34 | 1.23 |
| nC4 | 1.65 | 58.12 | 0.96 | 0.79 | 2.90 |
| iC5 | 0.73 | 72.15 | 0.53 | 0.44 | 1.61 |
| nC5 | 0.87 | 72.15 | 0.63 | 0.52 | 1.91 |
| C6 | 1.33 | 86.18 | 1.15 | 0.96 | 3.51 |
| C7+ | 36.11 | 254.86 | 92.03 | 82.31 | 301.80 |
| Total | 107.96 | 93.87 | 344.18 |
Table 1 Oil compositions and weight amount of CO2 produced by burning 1 mole of oil.
At reservoir conditions, CO2 and the oil in Table 1 will have approximately the same density. The CO2 formed by burning 1 reservoir cubic meter of oil will thus occupy a volume of around 3 cubic meters at reservoir conditions. As sketched in Figure 2, storing CO2 in depleted oil fields will therefore only provide space for one third of the CO2 produced. If the CO2 concentration in the atmosphere is not to increase, space must be found elsewhere for two thirds of the CO2 produced.
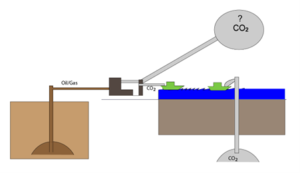
Figure 2 Illustration of the fact that depleted oil reservoirs can only hold a third of the CO2 produced by burning oil and gas.
CO2 capture
The oxygen (O2) needed for the combustion of gas and oil comes from atmospheric air. Table 2 shows the compositions of atmospheric air and of a typical combustion gas.
| Component | Atmospheric Air
Mol% |
Combustion Gas
Mol% |
| N2 | 78.08 | 66-71 |
| O2 | 20.95 | 2-3 |
| Ar | 0.93 | 0.7 |
| CO2 | 0.04 | 8-10 |
| H2O | 18-20 |
Table 2 Composition of atmospheric air and a typical combustion gas.
Before CO2 is disposed of, it must be separated from N2 and other gases. This can be done using an amine absorption process as sketched in Figure 3. CO2 is absorbed when the combustion gas contacts the amine (MEA) solution, while N2 and other gases are discharged at the top. The absorbed CO2 is separated from the amine solution in a heater.
The chemical reactions are:
Absorption:
MEA + CO2 → MEACOO– + H+
Bicarbonate formation:
MEACOO– + H2O → MEA + НСO–3
Desorption:
HCO–3 + H+ → CO2 + H2O
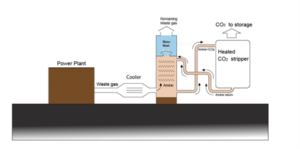
Figure 3 Amine absorption unit for separating CO2 from other gases contained in combustion gas.
CO2 at pipeline and storage conditions
Figure 4 shows the phase diagram of CO2. The CO2 molecule is linear, which in both the liquid and the vapor state favors a high density. The linear shape is also the reason CO2, compared to other components of approximately the same molecular weight, solidifies at a higher temperature.
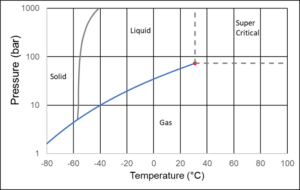
Figure 4 CO2 phase diagram.
Ship transport of CO2 will most often take place with CO2 placed in insolated cryogenic tanks, where the temperature is around -46°C and the pressure high enough to ensure that the CO2 is in liquid form, which means 8 bar or slightly higher. In a pipeline, the temperature will be determined by the ambient temperature, which will often be 4-30°C. The pressure is adjusted to ensure the density is high, without requiring an unreasonable compressor capacity. This is achieved at a pressure of 80-150 bar. Underground storage of CO2 in depleted oil fields or in aquifers could take place at a temperature of around 75°C and a pressure of around 225 bar.
Figure 5 shows the density of CO2 at typical shipping, pipeline and aquifer conditions. In marine transport, the CO2 density exceeds that of pure water, while at pipeline conditions the density is slightly lower, but still high considering that it is a condensed gas component. At aquifer conditions, CO2 is supercritical, but the density is still relatively high.
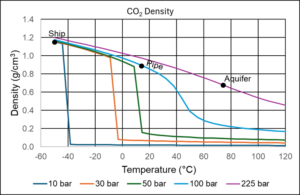
Figure 5 CO2 density at typical shipping, pipeline, and aquifer conditions.
Pipeline transport of CO2
In a pipeline, it is preferable to transport CO2 as single-phase liquid, but two-phase transport may occur. If so, it will be at temperature and pressure conditions located somewhere on the vapor pressure curve in Figure 4. This is a major difference compared to gas and oil mixtures, for which the two-phase region may cover a large PT area. This does not mean that the change from gas to liquid is instantaneous in a pipeline transporting CO2. Both gaseous and liquid CO2 can be present over long pipe distances, possibly in the entire pipe.
When carrying out flow simulations for a pipeline transporting gas and oil, it is for each section calculated how the heat (enthalpy) exchanged with the surroundings affects the relative phase quantities, their composition, and the temperature in the pipeline. This can either be done by table look-up or by continuously calculating how the phase quantities and compositions at the current pressure change with temperature. That method cannot be used for pure CO2. For a given pressure, the temperature remains fixed as long as two phases are present.
Figure 6 shows a Pressure-Enthalpy diagram for CO2. If two phases exist at a temperature of 10°C, the pressure is approximately 45 bar. The liquid phase will have an enthalpy of around -260 kJ/kg and the gas phase an enthalpy of around -63 kJ/kg. The total fluid will have an enthalpy in that range and the relative amounts of gas and liquid are determined using the lever rule.
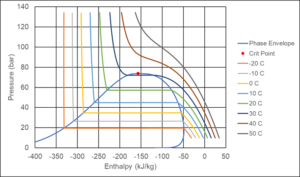
Figure 6 Pressure-Enthalpy (PH) diagram for CO2. The enthalpy reference state is ideal gas at 0°C.
Should a leak occur on a pipeline transporting natural gas, the leaking gas will rise into the air because the molecular weight of natural gas (18-20) is lower than that of atmospheric air (around 28.964). CO2 has a molecular weight of 44.0 and it will take time before the leaking CO2 mixes with the atmospheric air. In calm weather, CO2 from a leaking CO2 pipeline could form a kind of blanket around the leak and pose a hazard to people nearby.
Thermodynamic properties of pure and lightly polluted CO2
Thanks to the many industrial uses of CO2, good, validated, and easily accessible models exist for describing the properties of CO2. The volume-corrected SRK and PR equations are the industry standard for oil and gas and can also be used for CO2. In case more accurate CO2 properties are needed, one may use the Span-Wagner equation [1] developed for pure CO2, or the GERG-2008 equation [2]. The latter can handle mixtures of up to 20 specific components including CO2.
The CO2 to be transported and stored underground will seldom be pure CO2. Figure 7 shows the phase diagram of CO2 with trace amounts of nitrogen, argon, and water. It illustrates the fact that liquid water may well form in a pipeline transporting lightly polluted CO2. This may lead to corrosion problems as dealt with later. The industry standard SRK and PR equations are also applicable to mixtures with water, but it requires modifications capable of describing that water molecules when mixed with other components do not distribute randomly but prefer other water molecules as their nearest neighbors. Such modifications of the classical cubic equations are well described in the literature [3a].
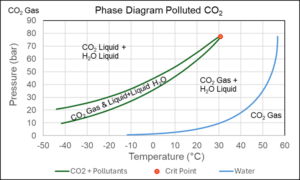
Figure 7 Phase diagram of mixture consisting of 97.7 mol% CO2, 1.6 mol% N2, 0.3 mol% Ar, and 0.4 mol% H2O.
Even trace amounts of water can lead to the formation of CO2 gas hydrate. This is a solid with properties similar to ice that can form at temperatures significantly higher than 0°C, which is the freezing point of water. Figure 8 shows a phase diagram for CO2 with trace amounts of water. As is seen from the figure, gas hydrate formation will only occur if some CO2 is in gaseous form, or the temperature is lower than around -13°C.
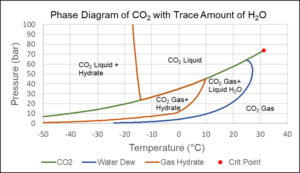
Figure 8 Phase diagram of mixture 99.9 mol% CO2 and 0.1 mol% H2O.
CO2 storage in depleted oil fields
The most obvious storage sites for the CO2 produced by burning oil and gas from petroleum reservoirs are the same reservoirs after the production by natural depletion has stopped. CO2 gas injection into oil fields has for many years been used as an enhanced oil recovery (EOR) technique. The injected CO2 mixes with the oil left in the depleted field and forms a zone that propagates through the field and pushes the oil forward towards the production well. It is a known and safe technique as long as the pressure in the reservoir does not exceed the original reservoir pressure. The mechanisms behind miscible gas displacement are well documented [3b]. Although this is an obvious storage option, additional storage space must be found because, as already mentioned, the CO2 produced takes up three times the volume that the produced oil or gas leaves in the reservoir.
CO2 storage in aquifers
The term Carbon Capture and Storage (CCS) was first used in the early 2000s. It was the time, it became clear that a new technical discipline was needed if virtually all CO2 from combustion gases were to be captured and purified, and sufficient storage space were to be found for the purified CO2. Brine reservoirs are present in large numbers worldwide and could be candidates to provide the additional storage capacity needed beyond that found in depleted oil fields.
The solubility of CO2 in saltwater is much lower than in oil. As illustrated in Figure 9 an aquifer volume of around 50 m3 would be needed if the CO2 produced by burning 1 reservoir cubic meter of oil were to dissolve in a brine aquifer [4].
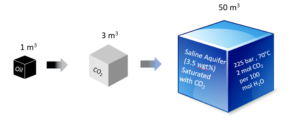
Figure 9 Volumetric development from oil at reservoir conditions to CO2 at reservoir conditions to CO2 dissolved in a brine aquifer.
According to the International Energy Agency, CO2 emissions in 2023 from the combustion of fossil fuels were 37.4 billion tons. If this much CO2 were to dissolve in a 200 m thick aquifer with a porosity of 20%, an aquifer area of the order of 20,500 square km would be needed. This is 47% of the area of Denmark. It seems unrealistic to find storage capacity in brine aquifers of that order of magnitude every year. Most of the CO2 stored in the brine aquifers will therefore remain in the form of free CO2 with some water content (a so-called CO2 plume). When assessing the storage capacity of a brine aquifer, permeability and mechanical strength of the rock material are therefore more important than the CO2 solubility in brine.
Risk of dissolution of rock material
Chalk and limestone are frequently occurring underground rock materials and consist of calcium carbonate (CaCO3). CO2 dissolved in water (H2O) can dissolve calcium carbonate by converting it to calcium and hydrogen carbonate ions:
CaCO3 + CO2 + H2O → Ca++ + 2HCO–3
Each mole of CO2 (44 g) that dissolves in water can dissolve one mole of calcium carbonate (100 g). If the production of CO2 in 2023 of 37.4 billion tons were injected into a limestone reservoir with a porosity of 20% and the size of Denmark (42,952 km2), it would lead to the land surface sinking by 0.9 meters.
The dissolution issue is not limited to chalk and limestone. Most sandstone has a high concentration of potassium feldspar (KAlSi3O8), which may also dissolve or at least corrode when contacted with CO2 rich water.
If CO2 is stored in a rock material whose main components are insoluble in CO2-rich water, there will not be a risk of collapse, but CaCO3 could be the material that binds the rock together. If the binding material dissolves, CO2 might leak to the atmosphere.
Corrosion problems in pipelines transporting CO2
Transport of CO2 carrying water can cause corrosion problems in an iron pipeline. Dissolved in water, CO2 will form carbonic acid (H2CO3), which can react with iron to form iron carbonate (FeCO3). If the FeCO3 layer adheres well to the iron surface, it can act as a physical barrier limiting further corrosion. Otherwise, the iron may react with the hydroxide ions (OH–) in the water to form iron hydroxide (Fe(OH)2), which in the presence of oxygen can be oxidized to iron oxide (Fe2O3), commonly known as rust.
CCS in a nutshell
Although CO2 is a well described component, the need has arisen for a new CO2 discipline called CCS. The reason is that the amount of CO2 that needs to be handled and disposed of is far larger than seen in any industrial use of CO2. The mass of CO2 produced is three times the mass of the burned gas and oil. CCS only makes sense if its energy consumption is significantly less than the energy obtained by burning the corresponding amount of fossil fuels. It will save money if the infrastructure and the simulation models that already exist for the transport and production of oil and gas can be reused. However, CO2 differs in several respects from oil or gas. The two-phase area of purified CO2 is much narrower than what the oil and gas flow and reservoir simulators are designed for. Dissolved in water, CO2 acts as an acid that can cause problems with corrosion in pipelines and lead to the dissolution of the rock formation in chalk and limestone aquifers with the risk of the ground sinking or CO2 escaping into the atmosphere.
References
[1] Span, R. and W. Wagner (1996), A New Equation of State for Carbon Dioxide Covering the Fluid Region from the Triple-Point Temperature to 1100 K at Pressures up to 800 MPa, J. Phys. Chem. Ref. Data 25, 1509.
[2] Kunz, O. and Wagner, W., The GERG-2008 Wide-Range Equation of State for Natural Gases and Other Mixtures: An Expansion of GERG-2004, Journal of Chem & Eng Data 57, 2012, 3032-3091.
[3] Pedersen, K.S., Christensen, P.L. and Shaikh, J.A., Phase Behavior of Petroleum Reservoir Fluids, 3rd Edition, Chapters 15[b] and 16[], Taylor & Francis, Boca Raton, FL, 2024.
[4] Pedersen, K.S. and Christensen, P.L., CO2 Solubility in Brine Aquifers – Volumetric Considerations, Paper OTC-34849-MS presented at the Offshore Technology Conference Asia, Kuala Lumpur, February 27 – March 1, 2024.
EoS Modelling with the Critical Point as Anchor Point
Summary
The phase behavior of a pure hydrocarbon is throughout the gas and liquid region essentially determined by the critical temperature and pressure of the component. Two recent Kapexy publications [1,2] show that the critical point also for a reservoir fluid exerts a large influence on the phase behavior of the fluid in the whole gas and liquid region. The critical point is so decisive for the behavior of a reservoir fluid that, in addition to the critical point, only a measured saturation point is needed to develop a reliable equation of state model. A measured critical point is rarely available for reservoir fluids, but the critical point of an oil can be calculated from the composition of the fluid. The same applies to the critical point of the C7+ fraction of a gas condensate. By utilizing this knowledge, the time spent developing equation of state models can be significantly reduced and performed with far less measured data than is standard today. There is the additional advantage that equation of state modeling can be performed as soon as a compositional analysis is available and does not have to await the full PVT study.
Theory
The famous Dutch scientist, van der Waals (vdW) presented the first cubic equation of state in 1873.
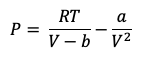
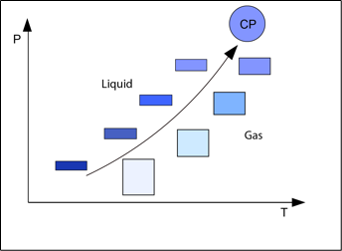
Figure 1 How van der Waals saw the development towards criticality for a pure hydrocarbon.
Today the industry standard cubic equations are the volume corrected Soave-Redlich-Kwong and Peng-Robinson equations. Unlike the van der Waals equation they can handle mixtures and therefore enable simulation of the critical point of a petroleum reservoir fluid.
Figure 2 shows the phase envelope of a reservoir fluid with iso-volume (quality) lines. The quality lines are bound to meet at the critical point, and the location of the critical point exerts much influence on the phase amounts, properties, and compositions also at pressures and temperatures far from the critical point.
Figure 2 Phase envelope of a petroleum reservoir fluid with critical point (CP) and iso-volume (quality) lines.
Given that the location of the critical point affects the phase behavior of a reservoir fluid throughout the gas and liquid regions, and that the critical point is the basis of the cubic equations, one may wonder why the critical point of a reservoir fluid is not assigned importance in equation of state (EoS) modeling. The focus is instead on matching PVT data (CME, CVD, Differential Liberation, etc.) measured at the reservoir temperature. A likely explanation is that the PVT studies that are standard today were defined at a time when it was not possible to simulate the critical point of a multicomponent mixture. Calculation of the critical point of multicomponent mixtures and flash calculations in the near-critical region only became possible with Professor Michael Michelsen’s pioneering work within phase equilibrium algorithms in the early 1980s [3].
EoS modeling consists of representing the C7+ fraction of a reservoir fluid as an appropriate number of pseudo-components and assigning values of Tc, Pc and acentric factor to each of the C7+ components. Petroleum reservoir fluids are not random mixtures. The logarithm of the mole percentages of the C7+ components develop linearly with carbon number. This pattern is the result of chemical reactions that have taken place over thousands of years. The critical temperature of the components contained in the C7+ fraction increases with carbon number and the critical pressure decreases. As illustrated in Figure 3, EoS modeling can therefore be simplified to rotating the curves representing the standard development of Tc and Pc with carbon number around a fixed value of C7 until the target PVT data is matched.
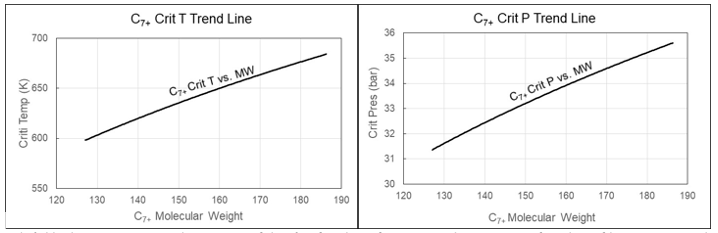
Figure 3 Development of Tc and Pc with carbon number for C7+ fractions. The full drawn lines show the default developments. The dashed lines show the variation accepted during EoS model development.
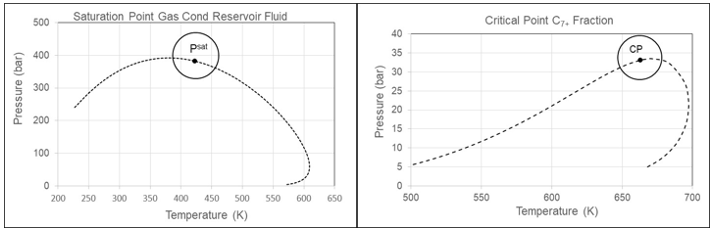
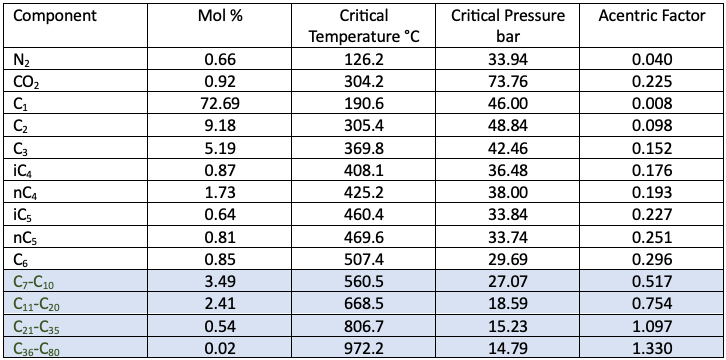
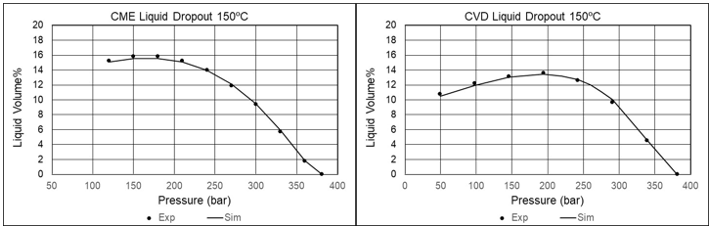
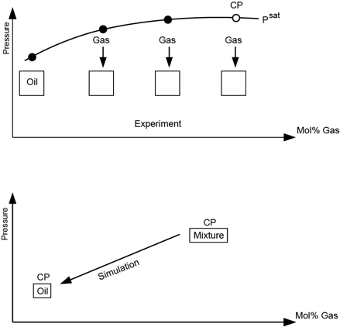
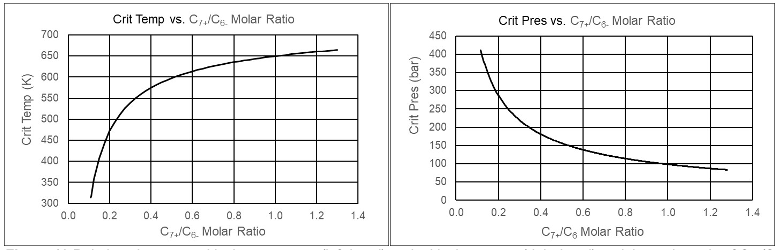
Gas Condensates
Those involved in equation of state (EoS) modeling for gas condensates will be familiar with the situation where the saturation point of the reservoir fluid is easily matched but the liquid dropout curves are not. The problem is exemplified in the left-hand side of Figure 4, which shows the experimental liquid dropout curve for a gas condensate as well as the liquid dropout curves simulated with three different EoS models, all of which match the saturation pressure. With EoS_1, the simulated liquid dropout curve is too flat. A perfect match is seen with EoS_2, while with EoS_3 the saturation pressure is simulated to be a bubble point instead of a dew point. The reason the liquid dropout curves differ is to be found in different simulated critical points. As can be seen from the phase envelope plots on the right-hand side of Figure 4, with EoS_1, the fluid is simulated to have a critical temperature much lower than the reservoir temperature. With EoS_2, the simulated critical temperature is still lower than the reservoir temperature, but not by much. Finally, the simulated critical temperature with EoS_3 is higher than the reservoir temperature. The simulated saturation point at the reservoir temperature is a bubble point and the fluid would erroneously be classified as an oil.
Figure 4 The left-hand side shows experimental and simulated liquid dropout data for a gas condensate fluid. The simulated liquid dropout curves are for three different EoS models. The right-hand side shows phase envelopes and critical points simulated using each of the three EoS-models.
Kapexy has performed an analysis of a large number of gas condensate EoS models, all of which provided a good match of liquid dropout data measured in constant mass expansion and constant volume depletion experiments [2]. As illustrated in Figure 5, the critical temperature and critical pressure of the C7+ fraction were found to increase with the average molecular weight.
Figure 5 Simulated phase envelopes for the C7+ fractions of two gas condensates exemplifying the trend that the critical temperature and the critical pressure increase with average molecular weight of the C7+ fraction.
Correlations were developed for predicting the critical point of the C7+ fraction of a gas condensate from its average molecular weight [2]. These correlations are graphically illustrated in Figure 6.
Figure 6 Critical temperature and pressure of the C7+ fraction of a gas condensate as a function of its average molecular weight.
For any gas condensate fluid, it is now possible from Figure 6 to read off the critical temperature and the critical pressure of the C7+ fraction based on its average molecular weight. If a measured saturation point exists for the entire fluid, the information in Figure 7 is available. Subsequently, the Tc and Pc curves in Figure 3 are rotated around the values for C7 until the simulation results agree with the data in Figure 7. The result can be as shown in Table 1 and as exemplified for this fluid in Figure 8, this will normally give a good agreement with the measured liquid dropout curves.
Figure 7 Data points deciding the phase behavior of the gas condensate reservoir fluid in Table 1.
Table 1 Gas condensate reservoir fluid for which the C7+ fraction has been characterized to match data in Figure 7. Experimental and simulated liquid dropout curves can be seen from Figure 8.
Figure 8 Experimental and simulated CME and CVD liquid dropout curves for the gas condensate mixture in Table 1, which has an average C7+ molecular weight of 165.
Oil Mixture
A swelling experiment stands out among PVT experiments. It is performed at the reservoir temperature and starts with a measurement of the saturation point of the reservoir oil. Gas is added in steps, and after each gas addition, the saturation pressure is measured. It is further recorded when the saturation point changes from a bubble point to a dew point. Between the last bubble point and the first dew point is a composition that is critical at the saturation pressure. If the equation of state parameters are tuned to match that this composition is critical, it allows the critical point of the reservoir oil to be determined as is illustrated in Figure 9. This is done using the same EoS model to simulate the critical point of the fluid after having removed the added gas. The components in the added gas have well-defined equation of state parameters, and the assumption is that these components are correctly described both in the critical fluid composition with gas added and in the reservoir fluid before adding any gas.
Figure 9 How to determine the critical point of a reservoir oil
Swelling experiments are quite costly and will rarely be conducted unless gas injection is considered as an increased recovery technique. Fortunately, sufficient swelling data has been published to allow correlations to be developed for predicting how the critical temperature and pressure of a reservoir oil evolve with the C7+/C6- hydrocarbon molar ratio. This development is shown graphically in Figure 10 and is valid for reservoir fluids for which at least 10 mol% of the hydrocarbons are C 7+ components.
Figure 10 Relations between critical temperature (left-hand) and critical pressure (right-hand) and the molar ratio of C7+/C6- hydrocarbons for a reservoir oil composition.
The EoS modeling procedure, utilizing the found relationship between the critical point and the C7+/C6- hydrocarbon molar ratio, is as follows for reservoir fluids for which at least 10 mol% of the hydrocarbons are C7+ components [1].
- Make a copy of the fluid and remove inorganics (N2, CO2 and H2S).
- Calculate the molar C7+/C6- hydrocarbon ratio for the fluid from 1.
- Read off from Figure 10 the critical temperature and pressure for the fluid from 1.
- Rotate the Tc and Pc curves in Figure 3 to match the measured saturation pressure of the reservoir fluid and the critical point from 3. of the reservoir fluid freed of inorganics.
- Use the resulting EoS model for the reservoir fluid. It is applicable at both reservoir and process conditions as well as when gas injection is used as an EOR technique.
References
[1] Pedersen, K.S., Shaikh, J.A. and Christensen, P.L., Importance of critical point for the phase behavior of a reservoir fluid, Fluid Phase Equilibria 573, 2023.
[2] Pedersen, K.S., Christensen, P.L. and Shaikh, J.A., Use of critical point in equation of state modeling for reservoir fluids, SPE-216776-MS, Abu Dhabi International Petroleum Exhibition & Conference, Abu Dhabi, October 2-5, 2023.
[3] Michelsen, M.L., Calculation of phase envelopes and critical points for multicomponent mixtures, Fluid Phase Equilibria 4, 1980, 1-10.
Contact
Find us: Firskovvej 36, 2800 Kgs. Lyngby
Send us an email: contact@kapexy.com
Call us: +45 88 77 29 02

Udviklet af ITAGIL